Abstract
Review Article
The effects of early low dose exposures to the Environmental Estrogen Bisphenol A on the Development of Childhood Asthma
Terumi Midoro-Horiuti* and Randall M Goldblum
Published: 10 July, 2017 | Volume 1 - Issue 1 | Pages: 015-027
Exposure to environmental chemicals is a potential cause for the rapid increase in the prevalence of allergic asthma over the last few decades. The production of the environmental estrogen bisphenol A, the monomer of polycarbonate plastics, has increased rapidly over the last 50 years, such that bisphenol A is one of the most highly produced chemicals. It is detectable in the urine of the vast majority of the human population. While the relationship between the increase of bisphenol A in our environment and the prevalence of asthma does not prove a cause and effect relationship, it provides a strong rationale for experiments that have tested the hypothesis. Because of its small molecular size and hydrophobicity, bisphenol A is easily transferred from the mother to the fetus, via the placenta and in breast milk.
We have reviewed all the publications available on medline on the human epidemiological studies of the early bisphenol A exposure on the development of allergic asthma and experimental studies using mouse model of the effects of early bisphenol A exposure on the development of asthma. There are eight human epidemiological studies and five mouse model studies currently published.
The human studies suggest that bisphenol A exposure in early life enhances the likelihood of developing asthma on at least one of the study groups. The effects of early bisphenol A exposure were observed as an enhanced development of asthma before adolescent in the animal model.
Read Full Article HTML DOI: 10.29328/journal.haard.1001003 Cite this Article Read Full Article PDF
References
- Kahr N, Naeser V, Stensballe LG, Kyvik KO, Skytthe A, et al. Gene-environment interaction in atopic diseases: a population-based twin study of early-life exposures. Clin Respir J. 2015; 9: 79-86. Ref.: https://goo.gl/LnKi1t
- Bonds RS, Midoro-Horiuti T. Estrogen effects in allergy and asthma. Curr Opin Allergy Clin Immunol. 2013; 13: 92-99. Ref.: https://goo.gl/1sEYTe
- Ng SC, Gilman-Sachs A, Thaker P, Beaman KD, Beer AE, et al. Expression of intracellular Th1 and Th2 cytokines in women with recurrent spontaneous abortion, implantation failures after IVF/ET or normal pregnancy. Am J Reprod Immunol. 2002; 48: 77-86. Ref.: https://goo.gl/edikvY
- Piccinni MP, Maggi E, Romagnani S. Role of hormone-controlled T-cell cytokines in the maintenance of pregnancy. Biochem Soc Trans. 2000; 28: 212-215. Ref.: https://goo.gl/3JnN5m
- Iwata M, Eshima Y, Kagechika H, Miyaura H. The endocrine disruptor’s nonylphenol and octylphenol exert direct effects on T cells to suppress Th1 development and enhance Th2 development. Immunol Lett. 2004; 94: 135-139. Ref.: https://goo.gl/gEpexT
- Yan H, Takamoto M, Sugane K. Exposure to Bisphenol A prenatally or in adulthood promotes T (H) 2 cytokine production associated with reduction of CD4CD25 regulatory T cells. Environ Health Perspect. 2008; 116: 514-519. Ref.: https://goo.gl/AQ3REY
- Cai Y, Zhou J, Webb DC. Estrogen stimulates Th2 cytokine production and regulates the compartmentalisation of eosinophils during allergen challenge in a mouse model of asthma. Int Arch Allergy Immunol. 2012; 158: 252-260. Ref.: https://goo.gl/q8F5EQ
- Zincke T. Mittheilungen aus dem chemischen Laboratorium der Universitat Marburg. Justus Leibigs Annals Chemie. 1905; 343: 75-99.
- Dodds Ec, Huang Rl, Lawson W, Robinson R. Synthetic oestrogenic compounds related to stilbene and diphenylethane. III. Proc R Soc Lond B Biol Sci. 1953; 140: 470-497. Ref.: https://goo.gl/bwPkuJ
- Akinbami LJ, Moorman JE, Liu X. Asthma prevalence, health care use, and mortality: United States, 2005-2009. Natl Health Stat Report. 2011; 1-14. Ref.: https://goo.gl/Vb5s66
- Tilson HA. Collection Bisohenol A (BPA). Env Health Perspectives. 2012; 1-37.
- Hines EP, Mendola P, von Ehrenstein OS, Ye X, Calafat AM, et al. Concentrations of environmental phenols and parabens in milk, urine and serum of lactating North Carolina women. Reprod Toxicol. 2015; 54: 120-128. Ref.: https://goo.gl/ahcDqZ
- Vandenberg LN, Hauser R, Marcus M, Olea N, Welshons WV. Human exposure to bisphenol A (BPA). Reprod Toxicol. 2007; 24: 139-177. Ref.: https://goo.gl/oZQTtA
- Lv Y, Lu S, Dai Y, Rui C, Wang Y, et al. Higher dermal exposure of cashiers to BPA and its association with DNA oxidative damage. Environ Int. 2017; 98: 69-74. Ref.: https://goo.gl/FfKhXY
- Hormann AM, Vom Saal FS, Nagel SC, Stahlhut RW, Moyer CL, et al. Holding thermal receipt paper and eating food after using hand sanitizer results in high serum bioactive and urine total levels of bisphenol A (BPA). PLoS One. 2014; 9: e110509. Ref.: https://goo.gl/DCiDgm
- Nakajima Y, Goldblum RM, Midoro-Horiuti T. Fetal exposure to bisphenol A as a risk factor for the development of childhood asthma: an animal model study. Environ Health. 2012; 11: 1-7. Ref.: https://goo.gl/Th2j3o
- Vinas R, Goldblum RM, Watson CS. Rapid estrogenic signaling activities of the modified (chlorinated, sulfonated, and glucuronidated) endocrine disruptor bisphenol A. Endocrine Disruptors. 2013; 1: e25411. Ref.: https://goo.gl/BWoQJ5
- Vandenberg LN, Chahoud I, Heindel JJ, Padmanabhan V, Paumgartten FJ, et al. Urinary, circulating, and tissue biomonitoring studies indicate widespread exposure to bisphenol A. Environ Health Perspect. 2010; 118: 1055-1070. Ref.: https://goo.gl/UDJAQk
- Vaidya SV, Kulkarni H. Association of Urinary Bisphenol A Concentration with Allergic Asthma: Results from the National Health and Nutrition Examination Survey 2005-2006. J Asthma. 2012; 49: 800-806. Ref.: https://goo.gl/4D6xWP
- Spanier AJ, Kahn RS, Kunselman AR, Hornung R, Xu Y, et al. Prenatal exposure to bisphenol a and child wheeze from birth to 3 years of age. Environ Health Perspect. 2012; 120: 916-920. Ref.: https://goo.gl/fCj5PZ
- Spanier AJ, Kahn RS, Kunselman AR, Schaefer EW, Hornung R, et al. Bisphenol A Exposure and the Development of Wheeze and Lung Function in Children Through Age 5 Years. JAMA Pediatr. 2014; 168: 1131-1137. Ref.: https://goo.gl/HkVeRi
- Donohue KM, Miller RL, Perzanowski MS, Just AC, Hoepner LA, et al. Prenatal and postnatal bisphenol A exposure and asthma development among inner-city children. J Allergy Clin Immunol. 2013; 131: 726-742. Ref.: https://goo.gl/rcN1Pc
- Kim K, Kim J, Kwon H, Hong S, Kim B, et al. Bisphenol A Exposure and Asthma Development in School-Age Children: A Longitudinal Study. PLOS One. 2014b; 9: e111383. Ref.: https://goo.gl/ab8Tus
- Whyatt RM, Rundle AG, Perzanowski MS, Just AC, Donohue KM, et al. Prenatal phthalate and early childhood bisphenol A exposures increase asthma risk in inner-city children. J Allergy Clin Immunol. 2014; 134: 1195-1197. Ref.: https://goo.gl/YJtU2y
- Gascon M, Casas M, Morales E, Valvi D, Ballesteros-Gomez A, et al. Prenatal exposure to bisphenol A and phthalates and childhood respiratory tract infections and allergy. J Allergy Clin Immunol. 2015; 135: 370-378. Ref.: https://goo.gl/NYK335
- Wang IJ, Chen CY, Bornehag CG. Bisphenol A exposure may increase the risk of development of atopic disorders in children. Int J Hyg Environ Health. 2016; 219: 311-316. Ref.: https://goo.gl/vm1CEs
- Kim JH, Sartor MA, Rozek LS, Faulk C, Anderson OS, et al. Perinatal bisphenol A exposure promotes dose-dependent alterations of the mouse methylome. BMC Genomics. 2014a; 15: 30. Ref.: https://goo.gl/G4fCRz
- Larsson K, Ljung BK, Palm B, Wennberg M, Kaj L, et al. Exposure determinants of phthalates, parabens, bisphenol A and triclosan in Swedish mothers and their children. Environ Int. 2014; 73: 323-333. Ref.: https://goo.gl/Tm5smM
- Covaci A, Hond ED, Geens T, Govarts E, Koppen G, et al. Urinary BPA measurements in children and mothers from six European member states: Overall results and determinants of exposure. Environ Res. 2015; 141: 77-85. Ref.: https://goo.gl/999Gex
- Midoro-Horiuti T, Tiwari R, Watson CS, Goldblum RM. Maternal exposure to bisphenol A enhances susceptibility to experimental allergic asthma in their pups. Env Health Perspectives. 2010; 118: 273-277.
- Bauer SM, Roy A, Emo J, Chapman TJ, Georas SN, et al. The effects of maternal exposure to bisphenol A on allergic lung inflammation into adulthood. Toxicol Sci. 2012; 130: 82-93. Ref.: https://goo.gl/rHdZxY
- Petzold S, Averbeck M, Simon JC, Lehmann I, Polte T. Lifetime-dependent effects of bisphenol A on asthma development in an experimental mouse model. PLoS One. 2014; 9: e100468. Ref.: https://goo.gl/7h8PLJ
- O'Brien E, Bergin IL, Dolinoy DC, Zaslona Z, Little RJ, et al. Perinatal bisphenol A exposure beginning before gestation enhances allergen sensitization, but not pulmonary inflammation, in adult mice. J Dev Orig Health Dis. 2014; 5: 121-131. Ref.: https://goo.gl/Qcr4zF
- Nygaard UC, Vinje NE, Samuelsen M, Andreassen M, Groeng EC, et al. Early life exposure to bisphenol A investigated in mouse models of airway allergy, food allergy and oral tolerance. Food Chem Toxicol. 2015; 83: 17-25. Ref.: https://goo.gl/E65agS
- Leme AS, Hubeau C, Xiang Y, Goldman A, Hamada K, et al. Role of breast milk in a mouse model of maternal transmission of asthma susceptibility. J Immunol. 2006; 176: 762-769. Ref.: https://goo.gl/4FnZaH
- Midoro-Horiuti T. Environmental estrogens and allergy. Japanese Journal of Pediatric Intaractable Asthma and Allergic Diseases. 2014; 12: 311-314.
- Bronson FH, Dagg CP, Snell GD. Reproduction. In: Green,E.L. (Ed.), Biology of the Laboratory Mouse. Dover Publications, INC. New York. 2007.
- Bachmanov AA, Reed DR, Beauchamp GK, Tordoff MG. Food intake, water intake, and drinking spout side preference of 28 mouse strains. Behav Genet. 2002; 32: 435-443. Ref.: https://goo.gl/SQPtVE
- Allam JP, Zivanovic O, Berg C, Gembruch U, Bieber T, et al. In search for predictive factors for atopy in human cord blood. Allergy. 2005; 60: 743-750. Ref.: https://goo.gl/Jcsbez
- Prescott SL, Macaubas C, Holt BJ, Smallacombe TB, Loh R, et al. Transplacental priming of the human immune system to environmental allergens: universal skewing of initial T cell responses toward the Th2 cytokine profile. J Immunol. 1998; 160: 4730-4737. Ref.: https://goo.gl/dV5Mpu
- Prescott SL, Macaubas C, Smallacombe T, Holt BJ, Sly PD, et al. Development of allergen-specific T-cell memory in atopic and normal children. Lancet. 1999; 353: 196-200. Ref.: https://goo.gl/Q6s9MX
- Gern JE, Lemanske RF Jr, Busse WW. Early life origins of asthma. J Clin Invest. 1999; 104: 837-843. Ref.: https://goo.gl/gvTu2v
- Holt PG, Jones CA. The development of the immune system during pregnancy and early life. Allergy. 2000; 55: 688-697. Ref.: https://goo.gl/unJVx8
- Strakovsky RS, Wang H, Engeseth NJ, Flaws JA, Helferich WG, et al. Developmental bisphenol A (BPA) exposure leads to sex-specific modification of hepatic gene expression and epigenome at birth that may exacerbate high-fat diet-induced hepatic steatosis. Toxicol Appl Pharmacol. 2015; 284: 101-112. Ref.: https://goo.gl/wiZny4
- Maccani MA, Marsit CJ. Exposure and fetal growth-associated miRNA alterations in the human placenta. Clin Epigenetics. 2011; 2: 401-404. Ref.: https://goo.gl/LiR6SY
- Dhimolea E, Wadia PR, Murray TJ, Settles ML, Treitman JD, et al. Prenatal exposure to BPA alters the epigenome of the rat mammary gland and increases the propensity to neoplastic development. PLoS One. 2014; 9: e99800. Ref.: https://goo.gl/wM6NP9
- Li G, Chang H, Xia W, Mao Z, Li Y, et al. F0 maternal BPA exposure induced glucose intolerance of F2 generation through DNA methylation change in Gck. Toxicol Lett. 2014; 228: 192-199. Ref.: https://goo.gl/Fj28CP
- Bastos SL, Kamstra JH, Cenijn PH, van Rijt LS, Hamers T, et al. Effects of endocrine disrupting chemicals on In Vitro global DNA methylation and adipocyte differentiation. Toxicol In Vitro. 2013; 27: 1634-1643. Ref.: https://goo.gl/5H6LWe
- Warita K, Mitsuhashi T, Ohta K, Suzuki S, Hoshi N, et al. Gene expression of epigenetic regulatory factors related to primary silencing mechanism is less susceptible to lower doses of bisphenol A in embryonic hypothalamic cells. J Toxicol Sci. 2013; 38: 285-289. Ref.: https://goo.gl/sCo2Hj
- Manikkam M, Tracey R, Guerrero-Bosagna C, Skinner MK. Plastics derived endocrine disruptors (BPA, DEHP and DBP) induce epigenetic transgenerational inheritance of obesity, reproductive disease and sperm epimutations. PLoS One. 2013; 8: e55387. Ref.: https://goo.gl/DSbpEx
- Doshi T, Mehta SS, Dighe V, Balasinor N, Vanage G. Hypermethylation of estrogen receptor promoter region in adult testis of rats exposed neonatally to bisphenol A. Toxicology. 2011; 289: 74-82. Ref.: https://goo.gl/8AFvsp
- Bromer JG, Zhou Y, Taylor MB, Doherty L, Taylor HS. Bisphenol-A exposure in utero leads to epigenetic alterations in the developmental programming of uterine estrogen response. FASEB J. 2010; 24: 2273-2280. Ref.: https://goo.gl/kiUQgb
- Doherty LF, Bromer JG, Zhou Y, Aldad TS, Taylor HS. In utero exposure to diethylstilbestrol (DES) or bisphenol-A (BPA) increases EZH2 expression in the mammary gland: an epigenetic mechanism linking endocrine disruptors to breast cancer. Horm Cancer. 2010; 1: 146-155. Ref.: https://goo.gl/ak8jsj
- Trapphoff T, Heiligentag M, El Hajj N, Haaf T, Eichenlaub-Ritter U. Chronic exposure to a low concentration of bisphenol A during follicle culture affects the epigenetic status of germinal vesicles and metaphase II oocytes. Fertil Steril. 2013; 100: 1758-1767. Ref.: https://goo.gl/4HikbX
- Doshi T, D'souza C, Dighe V, Vanage G. Effect of neonatal exposure on male rats to bisphenol A on the expression of DNA methylation machinery in the postimplantation embryo. J Biochem Mol Toxicol. 2012; 26: 337-343. Ref.: https://goo.gl/wWeH99
- Avissar-Whiting M, Veiga KR, Uhl KM, Maccani MA, Gagne LA, et al. Bisphenol A exposure leads to specific microRNA alterations in placental cells. Reprod Toxicol. 2010; 29: 401-406. Ref.: https://goo.gl/AyHLzH
- Dolinoy DC, Huang D, Jirtle RL. Maternal nutrient supplementation counteracts bisphenol A-induced DNA hypomethylation in early development. Proc Natl Acad Sci USA. 2007; 104: 13056-13061. Ref.: https://goo.gl/Sejh9t
- Yeo M, Berglund K, Hanna M, Guo JU, Kittur J, et al. Bisphenol A delays the perinatal chloride shift in cortical neurons by epigenetic effects on the Kcc2 promoter. Proc Natl Acad Sci USA. 2013; 110: 4315-4320. Ref.: https://goo.gl/TmygRn
- Yaoi T, Itoh K, Nakamura K, Ogi H, Fujiwara Y, et al. Genome-wide analysis of epigenomic alterations in fetal mouse forebrain after exposure to low doses of bisphenol A. Biochem Biophys Res Commun. 2008; 376: 563-567. Ref.: https://goo.gl/3BLehT
- Patella V, Giuliano A, Bouvet JP, Marone G. Endogenous superallergen protein Fv induces IL-4 secretion from human Fc epsilon RI+ cells through interaction with the VH3 region of IgE. J Immunol. 1998; 161: 5647-5655. Ref.: https://goo.gl/DxR4TP
- Kim JH, Rozek LS, Soliman AS, Sartor MA, Hablas A, et al. Bisphenol A-associated epigenomic changes in prepubescent girls: a cross-sectional study in Gharbiah, Egypt. Environ Health. 2013; 12: 12-33. Ref.: https://goo.gl/UBMptm
- Wong RL, Walker CL. Molecular pathways: environmental estrogens activate nongenomic signaling to developmentally reprogram the epigenome. Clin Cancer Res. 2013; 19: 3732-3737. Ref.: https://goo.gl/R7FR59
- Jeng YJ, Watson CS. Combinations of physiologic estrogens with xenoestrogens alter ERK phosphorylation profiles in rat pituitary cells. Environ Health Perspect. 2011; 119: 104-112. Ref.: https://goo.gl/boz1T3
- Chang LC, Lin HY, Tsai MT, Chou RH, Lee FY, et al. YC-1 inhibits proliferation of breast cancer cells by down regulating EZH2 expression via activation of c-Cbl and ERK. Br J Pharmacol. 2014; 171: 4010-4025. Ref.: https://goo.gl/gVLMmS
- Muller J, Hart CM, Francis NJ, Vargas ML, Sengupta A, et al. Histone methyltransferase activity of a Drosophila Polycomb group repressor complex. Cell. 2002; 111: 197-208. Ref.: https://goo.gl/iT9CyF
- Kuzmichev A, Nishioka K, Erdjument-Bromage H, Tempst P, Reinberg D. Histone methyltransferase activity associated with a human multiprotein complex containing the Enhancer of Zeste protein. Genes Dev. 2002; 16: 2893-2905. Ref.: https://goo.gl/jS6fmB
- Czermin B, Melfi R, McCabe D, Seitz V, Imhof A, et al. Drosophila enhancer of Zeste/ESC complexes have a histone H3 methyltransferase activity that marks chromosomal Polycomb sites. Cell. 2002; 111: 185-196. Ref.: https://goo.gl/mLmecg
- Cao R, Wang L, Wang H, Xia L, Erdjument-Bromage H, et al. Role of histone H3 lysine 27 methylation in Polycomb-group silencing. Science. 2002; 298: 1039-1043. Ref.: https://goo.gl/BuhaKb
- Lambert KC, Curran EM, Judy BM, Milligan GN, Lubahn DB, et al. Estrogen Receptor {alpha} (ER{alpha}) Deficiency in Macrophages Results in Increased Stimulation of CD4+ T Cells while 17{beta}-Estradiol Acts through ER{alpha} to Increase IL-4 and GATA-3 Expression in CD4+ T Cells Independent of Antigen Presentation. J Immunol. 2005; 175: 5716-5723. Ref.: https://goo.gl/nkpqej
- Zaitsu M, Narita S, Lambert KC, Grady JJ, Estes DM, et al. Estradiol activates mast cells via a non-genomic estrogen receptor-alpha and calcium influx. Mol Immunol. 2007; 44: 1987-1995. Ref.: https://goo.gl/s2idwm
- He S, Xie F, Liu Y, Tong Q, Mochizuki K, et al. The histone methyltransferase Ezh2 is a crucial epigenetic regulator of allogeneic T-cell responses mediating graft-versus-host disease. Blood. 2013; 122: 4119-4128. Ref.: https://goo.gl/4yG6zo
- Jacob E, Hod-Dvorai R, Schif-Zuck S, Avni O. Unconventional association of the polycomb group proteins with cytokine genes in differentiated T helper cells. J Biol Chem. 2008; 283: 13471-13481. Ref.: https://goo.gl/QDd14d
- Tumes DJ, Onodera A, Suzuki A, Shinoda K, Endo Y, et al. The polycomb protein Ezh2 regulates differentiation and plasticity of CD4(+) T helper type 1 and type 2 cells. Immunity. 2013; 39: 819-832. Ref.: https://goo.gl/EpeHw1
- Koyanagi M, Baguet A, Martens J, Margueron R, Jenuwein T, et al. EZH2 and histone 3 trimethyl lysine 27 associated with Il4 and Il13 gene silencing in Th1 cells. J Biol Chem. 2005; 280: 31470-31477. Ref.: https://goo.gl/HjZGog
- Bredfeldt TG, Greathouse KL, Safe SH, Hung MC, Bedford MT, et al. Xenoestrogen-induced regulation of EZH2 and histone methylation via estrogen receptor signaling to PI3K/AKT. Mol Endocrinol. 2010; 24: 993-1006. Ref.: https://goo.gl/2KbVSw
- Greathouse KL, Bredfeldt T, Everitt JI, Lin K, Berry T, et al. Environmental estrogens differentially engage the histone methyltransferase EZH2 to increase risk of uterine tumorigenesis. Mol Cancer Res. 2012; 10: 546-557. Ref.: https://goo.gl/ztTtxa
- Shimizu M, Ohta K, Matsumoto Y, Fukuoka M, Ohno Y, et al. Sulfation of bisphenol A abolished its estrogenicity based on proliferation and gene expression in human breast cancer MCF7 cells. Toxicol In Vitro. 2002; 16: 549-556. Ref.: https://goo.gl/tPxh8V
- Fedulov AV, Kobzik L. Allergy risk is mediated by dendritic cells with congenital epigenetic changes. Am J Respir Cell Mol Biol. 2011; 44: 285-292. Ref.: https://goo.gl/kUEGHz
- Hollingsworth JW, Maruoka S, Boon K, Garantziotis S, Li Z, et al. In utero supplementation with methyl donors enhances allergic airway disease in mice. J Clin Invest. 2008; 118: 3462-3469. Ref.: https://goo.gl/438pQB
- Liu J, Ballaney M, Al-alem U, Quan C, Jin X, et al. Combined inhaled diesel exhaust particles and allergen exposure alter methylation of T helper genes and IgE production in vivo. Toxicol Sci. 2008; 102: 76-81. Ref.: https://goo.gl/pCAKzp
Figures:
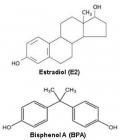
Figure 1
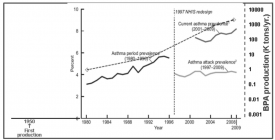
Figure 2
Similar Articles
-
Atopic Conjunctivitis in Children: Influence of Treatment with Topical Cyclosporin 0.05% in the Quality of LifeCarlos Alberto Sánchez Salguero*,Álvaro Isidro Sánchez Chacón. Atopic Conjunctivitis in Children: Influence of Treatment with Topical Cyclosporin 0.05% in the Quality of Life . . 2017 doi: 10.29328/journal.haard.1001001; 1: 001-008
-
Features of Interferon and Cytokine Status in Atopic DermatitisOspelnikova TP*,Gevorkyan OV, Mironova TV,Andreeva SA,Kolodyazhnaya LV,Ershov FI. Features of Interferon and Cytokine Status in Atopic Dermatitis. . 2017 doi: 10.29328/journal.haard.1001002; 1: 009-014
-
The effects of early low dose exposures to the Environmental Estrogen Bisphenol A on the Development of Childhood AsthmaTerumi Midoro-Horiuti*,Randall M Goldblum. The effects of early low dose exposures to the Environmental Estrogen Bisphenol A on the Development of Childhood Asthma. . 2017 doi: 10.29328/journal.haard.1001003; 1: 015-027
-
Prevalence of reported drug allergy and its impact on Beta lactam use with financial and health implicationsJoanna Lukawska*,Abirami Murugesh-Warre#,Ranu Malhin#,Yogini Jani,Christopher Corrigan,David Walker,Harsha Kariyawasam. Prevalence of reported drug allergy and its impact on Beta lactam use with financial and health implications. . 2017 doi: 10.29328/journal.haard.1001004; 1: 028-035
-
Inducible Laryngeal Obstruction/Vocal Cord Dysfunction and the Role It Plays in Refractory AsthmaJay I Peters*,Jorge Villalpando,Sandra G Adams. Inducible Laryngeal Obstruction/Vocal Cord Dysfunction and the Role It Plays in Refractory Asthma. . 2017 doi: 10.29328/journal.haard.1001005; 1: 036-039
-
The use of Allergoids and Adjuvants in Allergen ImmunotherapyCelso Eduardo Olivier*. The use of Allergoids and Adjuvants in Allergen Immunotherapy. . 2017 doi: 10.29328/journal.haard.1001006; 1: 040-060
-
Lack of applicability of the Enterocyte Chloride ion secretion paradigm to the Pathology of Cystic FibrosisMichael L Lucas*. Lack of applicability of the Enterocyte Chloride ion secretion paradigm to the Pathology of Cystic Fibrosis. . 2017 doi: 10.29328/journal.haard.1001007; 1: 061-085
-
Cytokine Modulatory Effects of Sesamum Indicum Seeds Oil Ameliorate Mice with Experimental Autoimmune EncephalomyelitisMohammad Reza Javan*,Mohammad Reza Zamani,Saeed Aslani,Ghader Dargahi Abbasabad,Masoud Beirami Khalaj,Hamed Serati-Nouri. Cytokine Modulatory Effects of Sesamum Indicum Seeds Oil Ameliorate Mice with Experimental Autoimmune Encephalomyelitis. . 2017 doi: 10.29328/journal.aaai.1001008; 1: 086-093
-
Chemo-cytokines network is main target for control of Allergic asthmaSeyyed Shamsadin Athari*,Seyyede Masoume Athari. Chemo-cytokines network is main target for control of Allergic asthma. . 2018 doi: 10.29328/journal.aaai.1001009; 2: 001-002
-
Role of Serum Magnesium levels in Asthmatic with childrenSomashekar AR*,Ramakrishnan KG,Seyyed Vanitha Gowda. Role of Serum Magnesium levels in Asthmatic with children. . 2018 doi: 10.29328/journal.aaai.1001010; 2: 003-005
Recently Viewed
-
Circulating platelet-derived vesicle in atrial fibrillationAlexander E Berezin*,Alexander A Berezin. Circulating platelet-derived vesicle in atrial fibrillation. Ann Clin Hypertens. 2019: doi: 10.29328/journal.ach.1001016; 3: 031-038
-
Hypertension as a persistent public health problem. A position paper from Alliance for a Healthy Heart, MexicoMartin Rosas-Peralta*,Luis Alcocer,Humberto Álvarez-López,Gabriela Borrayo-Sánchez,Ernesto Germán Cardona-Muñoz,Adolfo Chávez-Mendoza,Enrique Díaz y Díaz,José Manuel Enciso-Muñoz,Héctor Galván-Oseguera,Enrique Gómez-Álvarez,Pedro Gutiérrez-Fajardo,Héctor Hernández y Hernández,Francisco Javier León-Hernández,José Antonio Magaña-Serrano,José Zacarías Parra-Carrillo. Hypertension as a persistent public health problem. A position paper from Alliance for a Healthy Heart, Mexico. Ann Clin Hypertens. 2019: doi: 10.29328/journal.ach.1001015; 3: 009-030
-
Heart Failure with preserved Ejection Fraction (HFpEF); A Mexican cohort from Mexican Institute of Social Security (IMSS)Martin Rosas Peralta*,Lizette Arizmendi-Ocampo,Daniela Salinas Morales,Alicia Contreras Rodríguez,Erick Ramírez Arias,Carlos Riera kinkel,Guadalupe Castro Martínez,José Antonio Magaña-Serrano, Alejandro Chanona Espinosa. Heart Failure with preserved Ejection Fraction (HFpEF); A Mexican cohort from Mexican Institute of Social Security (IMSS). Ann Clin Hypertens. 2019: doi: 10.29328/journal.ach.1001014; 3: 001-008
-
Cannabinoids, Oxidative Stress and Female Reproduction: Where do we stand?Somenath Ghosh*. Cannabinoids, Oxidative Stress and Female Reproduction: Where do we stand?. Ann Adv Chem. 2025: doi: 10.29328/journal.aac.1001056; 9: 019-026
-
Strategic Plans for Diagnosis, Treatment & Control of HypertensionM Mohsen Ibrahim*. Strategic Plans for Diagnosis, Treatment & Control of Hypertension. Ann Clin Hypertens. 2018: doi: 10.29328/journal.ach.1001013; 2: 067-078
Most Viewed
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Evaluation of In vitro and Ex vivo Models for Studying the Effectiveness of Vaginal Drug Systems in Controlling Microbe Infections: A Systematic ReviewMohammad Hossein Karami*, Majid Abdouss*, Mandana Karami. Evaluation of In vitro and Ex vivo Models for Studying the Effectiveness of Vaginal Drug Systems in Controlling Microbe Infections: A Systematic Review. Clin J Obstet Gynecol. 2023 doi: 10.29328/journal.cjog.1001151; 6: 201-215
-
Prospective Coronavirus Liver Effects: Available KnowledgeAvishek Mandal*. Prospective Coronavirus Liver Effects: Available Knowledge. Ann Clin Gastroenterol Hepatol. 2023 doi: 10.29328/journal.acgh.1001039; 7: 001-010
-
Causal Link between Human Blood Metabolites and Asthma: An Investigation Using Mendelian RandomizationYong-Qing Zhu, Xiao-Yan Meng, Jing-Hua Yang*. Causal Link between Human Blood Metabolites and Asthma: An Investigation Using Mendelian Randomization. Arch Asthma Allergy Immunol. 2023 doi: 10.29328/journal.aaai.1001032; 7: 012-022
-
An algorithm to safely manage oral food challenge in an office-based setting for children with multiple food allergiesNathalie Cottel,Aïcha Dieme,Véronique Orcel,Yannick Chantran,Mélisande Bourgoin-Heck,Jocelyne Just. An algorithm to safely manage oral food challenge in an office-based setting for children with multiple food allergies. Arch Asthma Allergy Immunol. 2021 doi: 10.29328/journal.aaai.1001027; 5: 030-037

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."