Abstract
Research Article
Treatment protocol with alternative iron drugs in patients with an allergic reaction during iron replacement therapy
Saltuk Buğra Kaya*
Published: 01 February, 2023 | Volume 7 - Issue 1 | Pages: 001-004
In our study, we aimed to show that alternative iron salts containing different additives are safe to use in patients who have type 1 hypersensitivity reactions to iron drugs and need iron replacement therapy.
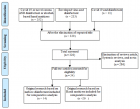
Materials and methods: Between January 2022 and June 2022, patients who had previously developed type 1 hypersensitivity reactions with iron preparations and needed iron replacement were included in the study. The study was designed retrospectively. Skin tests were first performed on patients to demonstrate a type 1 hypersensitivity reaction. If skin tests were negative and there was no history of life-threatening anaphylaxis, oral provocation tests were continued. If the absence of variability in symptoms and perimeter values, the drug allergy test was considered negative.
Results: Twenty-two patients were included in the study. Twenty-one of the patients were female and one was male. Iron deficiency anemia was found in nine patients, and low iron stores in thirteen patients without anemia were found. Type 1 hypersensitivity reaction developed with Iron 3 Carboxymaltose in 7 patients, Iron 2 Sulfate in 5 patients, Iron 2 Glycine in 4 patients, Iron 3 Hydroxy Polymaltose in 4 patients, Iron 2 Fumarate in 1 patient and Iron 3 Hydroxide Sucrose in 1 patient. Allergy tests with all alternative iron drugs containing additional additives were negative.
Conclusion: If patients with allergic reactions cannot be referred to allergy clinics, we think that oral iron salts with different additives can be used after the first dose is given in the hospital under general anaphylaxis precautions. We show that oral iron salts containing different additives can be safely used.
Read Full Article HTML DOI: 10.29328/journal.aaai.1001030 Cite this Article Read Full Article PDF
References
- Philip KEJ, Sadaka AS, Polkey MI, Hopkinson NS, Steptoe A, Fancourt D. The prevalence and associated mortality of non-anaemic iron deficiency in older adults: a 14 years observational cohort study. Br J Haematol. 2020 May;189(3):566-572. doi: 10.1111/bjh.16409. Epub 2020 Feb 18. PMID: 32072619; PMCID: PMC7613129.
- Tolkien Z, Stecher L, Mander AP, Pereira DI, Powell JJ. Ferrous sulfate supplementation causes significant gastrointestinal side-effects in adults: a systematic review and meta-analysis. PLoS One. 2015 Feb 20;10(2):e0117383. doi: 10.1371/journal.pone.0117383. PMID: 25700159; PMCID: PMC4336293.
- Auerbach M, Deloughery T. Single-dose intravenous iron for iron deficiency: a new paradigm. Hematology Am Soc Hematol Educ Program. 2016 Dec 2;2016(1):57-66. doi: 10.1182/asheducation-2016.1.57. PMID: 27913463; PMCID: PMC6142502.
- Rampton D, Folkersen J, Fishbane S, Hedenus M, Howaldt S, Locatelli F, Patni S, Szebeni J, Weiss G. Hypersensitivity reactions to intravenous iron: guidance for risk minimization and management. Haematologica. 2014 Nov;99(11):1671-6. doi: 10.3324/haematol.2014.111492. PMID: 25420283; PMCID: PMC4222472.
- de Barrio M, Fuentes V, Tornero P, Sánchez I, Zubeldia J, Herrero T. Anaphylaxis to oral iron salts. desensitization protocol for tolerance induction. J Investig Allergol Clin Immunol. 2008;18(4):305-8. PMID: 18714540.
- Yilmaz O, Erkasar Citak F. A case report with hypersensitivity to ferrous but tolerance of ferric iron salts. J Allergy Clin Immunol Pract. 2016 Mar-Apr;4(2):343-4.e1. doi: 10.1016/j.jaip.2015.11.001. Epub 2015 Nov 25. PMID: 26626063.
- Rodríguez-Jiménez B, Domínguez-Ortega J, Nuñez-Acevedo B, Cava-Sumner B, Kindelan-Recarte C, Montojo-Guillén C. Rapid iron desensitization after generalized urticaria and facial angioedema. J Investig Allergol Clin Immunol. 2014;24(1):69-71. PMID: 24765889.
- Özdemir Ö, Büyükavcı M. Development of hypersensitivity reactions after using different oral iron preparations. 2018.
- Bonadonna P, Brockow K. Drug Hypersensitivity and Clonal Mast Cell Disorders. In Cutaneous Drug Hypersensitivity. Springer, Cham. 2022; 161-165.
- Buonomo A, Nucera E, Criscuolo M. Treatment of Indolent and Advanced Systemic Mastocytosis. Mediterr J Hematol Infect Dis. 2022 May 1;14(1):e2022040. doi: 10.4084/MJHID.2022.040. PMID: 35615325; PMCID: PMC9084172.
Figures:
Similar Articles
-
Atopic Conjunctivitis in Children: Influence of Treatment with Topical Cyclosporin 0.05% in the Quality of LifeCarlos Alberto Sánchez Salguero*,Álvaro Isidro Sánchez Chacón. Atopic Conjunctivitis in Children: Influence of Treatment with Topical Cyclosporin 0.05% in the Quality of Life . . 2017 doi: 10.29328/journal.haard.1001001; 1: 001-008
-
Features of Interferon and Cytokine Status in Atopic DermatitisOspelnikova TP*,Gevorkyan OV, Mironova TV,Andreeva SA,Kolodyazhnaya LV,Ershov FI. Features of Interferon and Cytokine Status in Atopic Dermatitis. . 2017 doi: 10.29328/journal.haard.1001002; 1: 009-014
-
The effects of early low dose exposures to the Environmental Estrogen Bisphenol A on the Development of Childhood AsthmaTerumi Midoro-Horiuti*,Randall M Goldblum. The effects of early low dose exposures to the Environmental Estrogen Bisphenol A on the Development of Childhood Asthma. . 2017 doi: 10.29328/journal.haard.1001003; 1: 015-027
-
Prevalence of reported drug allergy and its impact on Beta lactam use with financial and health implicationsJoanna Lukawska*,Abirami Murugesh-Warre#,Ranu Malhin#,Yogini Jani,Christopher Corrigan,David Walker,Harsha Kariyawasam. Prevalence of reported drug allergy and its impact on Beta lactam use with financial and health implications. . 2017 doi: 10.29328/journal.haard.1001004; 1: 028-035
-
Inducible Laryngeal Obstruction/Vocal Cord Dysfunction and the Role It Plays in Refractory AsthmaJay I Peters*,Jorge Villalpando,Sandra G Adams. Inducible Laryngeal Obstruction/Vocal Cord Dysfunction and the Role It Plays in Refractory Asthma. . 2017 doi: 10.29328/journal.haard.1001005; 1: 036-039
-
The use of Allergoids and Adjuvants in Allergen ImmunotherapyCelso Eduardo Olivier*. The use of Allergoids and Adjuvants in Allergen Immunotherapy. . 2017 doi: 10.29328/journal.haard.1001006; 1: 040-060
-
Lack of applicability of the Enterocyte Chloride ion secretion paradigm to the Pathology of Cystic FibrosisMichael L Lucas*. Lack of applicability of the Enterocyte Chloride ion secretion paradigm to the Pathology of Cystic Fibrosis. . 2017 doi: 10.29328/journal.haard.1001007; 1: 061-085
-
Cytokine Modulatory Effects of Sesamum Indicum Seeds Oil Ameliorate Mice with Experimental Autoimmune EncephalomyelitisMohammad Reza Javan*,Mohammad Reza Zamani,Saeed Aslani,Ghader Dargahi Abbasabad,Masoud Beirami Khalaj,Hamed Serati-Nouri. Cytokine Modulatory Effects of Sesamum Indicum Seeds Oil Ameliorate Mice with Experimental Autoimmune Encephalomyelitis. . 2017 doi: 10.29328/journal.aaai.1001008; 1: 086-093
-
Chemo-cytokines network is main target for control of Allergic asthmaSeyyed Shamsadin Athari*,Seyyede Masoume Athari. Chemo-cytokines network is main target for control of Allergic asthma. . 2018 doi: 10.29328/journal.aaai.1001009; 2: 001-002
-
Role of Serum Magnesium levels in Asthmatic with childrenSomashekar AR*,Ramakrishnan KG,Seyyed Vanitha Gowda. Role of Serum Magnesium levels in Asthmatic with children. . 2018 doi: 10.29328/journal.aaai.1001010; 2: 003-005
Recently Viewed
-
Transcriptome-wide association study: Opportunity and challenges for cancer studiesKendy Shang,Jingni He*. Transcriptome-wide association study: Opportunity and challenges for cancer studies. Insights Biol Med. 2022: doi: 10.29328/journal.ibm.1001023; 6: 017-021
-
Pharmacological Manipulation of the Aging Pathways to Effect Health Span and Lifespan with Special Reference to SGLT2 Inhibitors as Powerful Anti-aging Agents in HumansDavid K Murdock*. Pharmacological Manipulation of the Aging Pathways to Effect Health Span and Lifespan with Special Reference to SGLT2 Inhibitors as Powerful Anti-aging Agents in Humans. Insights Biol Med. 2024: doi: 10.29328/journal.ibm.1001028; 8: 011-025
-
The motivational factors and adverse events experienced by healthy volunteers donating bone marrow for researchMirella Ejiugwo,Georgina Shaw,Frank Barry,Janusz Krawczyk,Veronica McInerney*. The motivational factors and adverse events experienced by healthy volunteers donating bone marrow for research. Int J Bone Marrow Res. 2019: doi: 10.29328/journal.ijbmr.1001010; 2: 089-096
-
Unveiling the Impostor: Pulmonary Embolism Presenting as Pneumonia: A Case Report and Literature ReviewSaahil Kumar,Karuna Sree Alwa*,Mahesh Babu Vemuri,Anumola Gandhi Ganesh Gupta,Nuthan Vallapudasu,Sunitha Geddada. Unveiling the Impostor: Pulmonary Embolism Presenting as Pneumonia: A Case Report and Literature Review. J Pulmonol Respir Res. 2025: doi: 10.29328/journal.jprr.1001065; 9: 001-005
-
Use of Lactobacillus reuteri DSM 17938 in the treatment of Stage II-III Periodontitis: Longitudinal Study of 36 PatientsAlessandra Laforgia*, Daniela Di Venere, Saverio Capodiferro, Vanja Granberg, Giuseppe Barile, Massimo Corsalini. Use of Lactobacillus reuteri DSM 17938 in the treatment of Stage II-III Periodontitis: Longitudinal Study of 36 Patients. J Clin Adv Dent. 2024: doi: 10.29328/journal.jcad.1001039; 8: 001-008
Most Viewed
-
Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth EnhancersH Pérez-Aguilar*, M Lacruz-Asaro, F Arán-Ais. Evaluation of Biostimulants Based on Recovered Protein Hydrolysates from Animal By-products as Plant Growth Enhancers. J Plant Sci Phytopathol. 2023 doi: 10.29328/journal.jpsp.1001104; 7: 042-047
-
Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case PresentationJulian A Purrinos*, Ramzi Younis. Sinonasal Myxoma Extending into the Orbit in a 4-Year Old: A Case Presentation. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001099; 8: 075-077
-
Feasibility study of magnetic sensing for detecting single-neuron action potentialsDenis Tonini,Kai Wu,Renata Saha,Jian-Ping Wang*. Feasibility study of magnetic sensing for detecting single-neuron action potentials. Ann Biomed Sci Eng. 2022 doi: 10.29328/journal.abse.1001018; 6: 019-029
-
Pediatric Dysgerminoma: Unveiling a Rare Ovarian TumorFaten Limaiem*, Khalil Saffar, Ahmed Halouani. Pediatric Dysgerminoma: Unveiling a Rare Ovarian Tumor. Arch Case Rep. 2024 doi: 10.29328/journal.acr.1001087; 8: 010-013
-
Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative reviewKhashayar Maroufi*. Physical activity can change the physiological and psychological circumstances during COVID-19 pandemic: A narrative review. J Sports Med Ther. 2021 doi: 10.29328/journal.jsmt.1001051; 6: 001-007

HSPI: We're glad you're here. Please click "create a new Query" if you are a new visitor to our website and need further information from us.
If you are already a member of our network and need to keep track of any developments regarding a question you have already submitted, click "take me to my Query."